In the highly regulated landscape of industrial manufacturing, medium and small-sized boilers and kilns present a unique engineering paradox. They require the same ultra-low emission standards as massive utility power plants, yet they must operate within severely constrained spatial footprints and strict capital expenditure limits. Traditional wet scrubbing systems—with their colossal water demands, heavy mechanical infrastructure, and wastewater treatment requirements—are fundamentally incompatible with these lean environments. Enter the Sodium Bicarbonate Dry Desulfurization (SDS) system. By leveraging high-temperature thermal activation and sub-micron pulverization, this entirely dry process achieves over 95 percent sulfur removal efficiency without generating a single drop of liquid effluent. This technical analysis explores the aerodynamic precision, chemical kinetics, and multi-pollutant collaborative control that make SDS the definitive solution for modern compact industrial operations.

Fig 1: Compact Industrial Integration of the BLSDS Series Dry Desulfurization Architecture
1. The Space-Constrained Imperative
The BLSDS series is engineered explicitly to dominate the mid-market industrial sector. Medium and small industrial boilers, metallurgy kilns, and glass furnaces are frequently located in dense, mature industrial parks where expanding the facility’s footprint is geographically impossible. The SDS process requires no absorption towers, no slurry circulation tanks, and no complex dewatering centrifuges. Instead, the reaction occurs dynamically within the flue duct and a specialized dry reactor, drastically minimizing the spatial requirement.
Operational Parameters
Despite its compact footprint, the system is an industrial powerhouse. It effortlessly manages gas volumes ranging from 10,000 to 2,300,000 cubic meters per hour. It thrives in high-temperature environments, permitting inlet temperatures up to 260 degrees Celsius. The aerodynamic design of the injection components ensures an operating resistance of merely 800 to 1000 Pa, allowing the facility to process inlet dust densities up to 1500 milligrams per normal cubic meter while reliably discharging purified air below the strict 35 milligrams per normal cubic meter threshold.
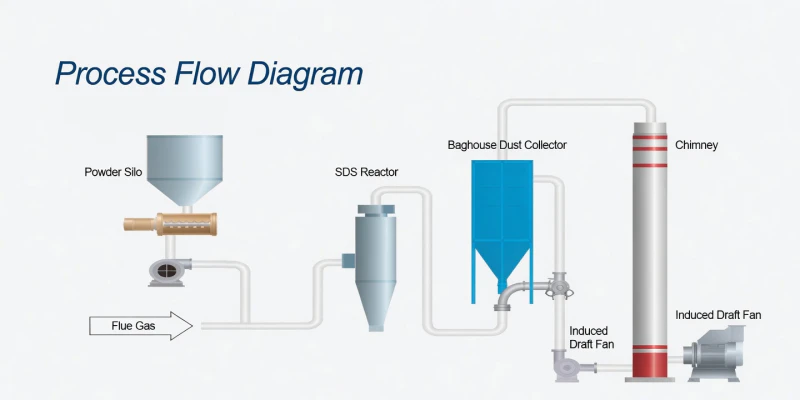
Fig 2: Holistic Process Flow: From Flue Gas Inlet to Baghouse Filtration
2. Thermal Activation and Solid-Phase Kinetics
The core brilliance of the SDS method lies in exploiting the thermal energy of the untreated flue gas to trigger an instantaneous chemical metamorphosis in the sorbent material.
The Popcorn Effect: Generating Micro-Pores
When hot flue gas (ranging from 140 to 260 degrees Celsius) enters the SDS reactor, ultra-fine sodium bicarbonate powder is pneumatically injected into the turbulent stream. Under the action of this high-temperature heat, the sodium bicarbonate undergoes a rapid endothermic decomposition. It breaks down into highly active sodium carbonate and carbon dioxide gas. As the carbon dioxide escapes the particle structure, it creates microscopic fissures and pores—a phenomenon colloquially known as the “popcorn effect.”
This newly formed, highly porous sodium carbonate possesses an immense specific surface area. It immediately and violently reacts with sulfur dioxide in the gas stream to form solid sodium sulfite, capturing the acidic pollutant in a dry, stable phase.
Synergistic Acid Mist Elimination
Beyond primary desulfurization, the highly active sodium carbonate also targets trace amounts of sulfur trioxide. By neutralizing this compound into sodium sulfate, the system eliminates the formation of highly corrosive sulfuric acid mist. This vital side reaction protects all downstream ductwork, fans, and stack infrastructure from catastrophic acid dew-point corrosion, dramatically extending the facility’s operational lifespan.
Primary Chemical Reactions
1. Thermal Decomposition:
2NaHCO₃ + Haba → Na₂CO₃ + CO₂↑ + H₂O
2. Main Desulfurization:
Na₂CO₃ + SO₂ → Na₂SO₃ + CO₂↑
3. Oxidation Side Reaction:
2Na₂SO₃ + O₂ → 2Na₂SO₄
4. Acid Mist Elimination:
Na₂CO₃ + SO₃ → Na₂SO₄ + CO₂↑
3. Micro-Engineering: The Pulverizing and Injection Matrix
To achieve a 95 percent plus desulfurization efficiency in a dry state, the physical particle size of the absorbent is the determining factor. Standard industrial sodium bicarbonate is far too coarse for instantaneous reaction. BAOLAN integrates an advanced classifying mill directly into the feed line to address this.
Sub-Micron Grinding and Transport
The classifying mill grinds domestic baking soda with a high crushing ratio and excellent energy utilization rate, achieving a powder output fineness exceeding 1000 mesh (particles smaller than 15 micrometers). This ultra-fine consistency guarantees that the mass transfer resistance between the solid particles and the flue gas is minimized.
Once pulverized, the material is handled by an automated vacuum feeding system. This sealed, pneumatic transport network ensures low labor intensity for operators and prevents fugitive dust from entering the factory environment. The ultra-fine powder is then propelled into the flue duct via proprietary SDS injection components. These injection lances create strategic aerodynamic turbulence, maximizing mixing uniformity and ensuring the gas-solid contact time exceeds the critical 4-second threshold required for total reaction.
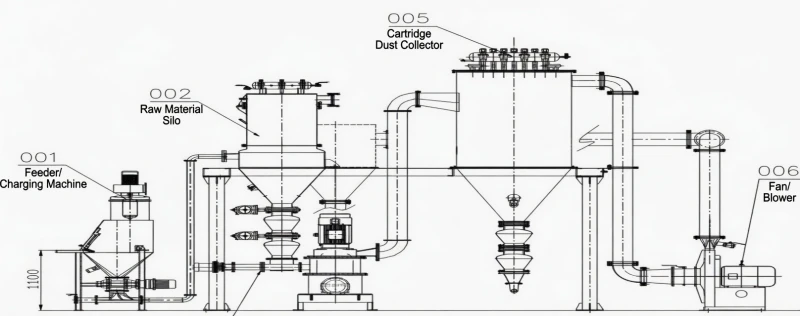
Fig 3: Automated Sub-Micron Pulverizing and Pneumatic Injection System
4. The Paradigm Shift: Engineering by Elimination
The true genius of the SDS Dry Desulfurization system lies not just in what it adds, but in what it completely obsoletes. Traditional wet and semi-dry desulfurization methods rely heavily on massive mechanical infrastructure to manage liquid slurries. By transitioning to a purely dry, gas-solid reaction, the SDS process eradicates the need for high-maintenance wet components, drastically lowering capital expenditure, reducing electrical load, and eliminating the risk of mechanical failure in hostile chemical environments.

Obsoleting Slurry Agitation
Wet scrubbers require colossal circulation tanks fitted with heavy-duty mechanical agitators to prevent the heavy calcium sulfite slurry from settling and turning into concrete-like scale. Because the SDS process utilizes lightweight, dry sodium bicarbonate powder suspended directly in the air stream, the entire liquid reservoir and its power-hungry agitators are completely engineered out of the equation.

Bypassing Forced Oxidation
In traditional limestone-gypsum systems, massive Roots blowers are forced to run continuously, pumping vast volumes of air into the liquid tanks to oxidize sulfites into stable sulfates. The SDS method utilizes the innate thermal energy and oxygen already present in the hot flue gas to achieve oxidation naturally. This bypasses the massive electrical load and noise pollution associated with forced aeration systems.
Eradicating Aerosol Demisting
Liquid spray towers generate dense, saturated mists that mandate complex, multi-stage corrugated demisters to prevent acid rain from escaping the stack. The SDS process generates absolutely zero liquid moisture. The flue gas remains completely dry, permanently eradicating the need for demisting infrastructure, eliminating winter plume opacity, and preventing downstream duct corrosion.
5. Multi-Pollutant Collaborative Control
The Filter Cake Synergistic Reaction
Following the primary dynamic reaction within the SDS duct, the gas stream—now carrying the newly formed solid sodium sulfate, original fly ash, and trace amounts of unreacted sodium carbonate—is directed into a high-temperature bag filter system. The filter material is engineered with specialized fibers capable of continuously withstanding temperatures exceeding 260 degrees Celsius without thermal degradation.
As the particulate matter collects on the surface of the filter bags, it forms a dense, alkaline “filter cake.” As the remaining flue gas is forced through this porous crust, any residual sulfur dioxide is subjected to a secondary, stationary chemical reaction. This synergistic process ensures that reagent utilization is maximized, operational costs are minimized, and multiple pollutants—including sulfur, dust, and acidic halides—are simultaneously stripped from the air stream before it ever reaches the induction fan.
Elevate Your Emission Control Standards Today
Don’t let the spatial limitations of your boiler room or the high maintenance demands of wet scrubbing systems compromise your facility’s environmental compliance. Implement the power of the BAOLAN SDS Dry Desulfurization system to achieve 95 percent plus efficiency, eliminate wastewater management, and ensure completely dry, plume-free operation. Contact our senior technical engineering team today to design a compact, automated dry-injection architecture custom-tailored to your exact industrial requirements.
