In the complex ecosystem of Volatile Organic Compound (VOC) abatement, the catalytic oxidizer acts as the terminal destination for molecular destruction. While advanced zeolite molecular sieves perform the vital function of concentrating dilute waste streams into energy-rich airflows, it is the catalyst—rightfully termed the Heart of the system—that executes the definitive chemical transformation. By systematically lowering the activation energy of oxidation reactions, these specialized materials convert hazardous solvents into harmless water vapor and carbon dioxide. In high-stakes environments such as semiconductor fabrication, pharmaceutical synthesis, and industrial printing, a catalyst is subjected to brutal thermal cycles and aerodynamic forces. To ensure a 95 percent plus purification rate and long-term economic viability, a catalyst must possess four uncompromising pillars of excellence: High Activity, Thermal Stability, Mechanical Strength, and an Extended Service Life.
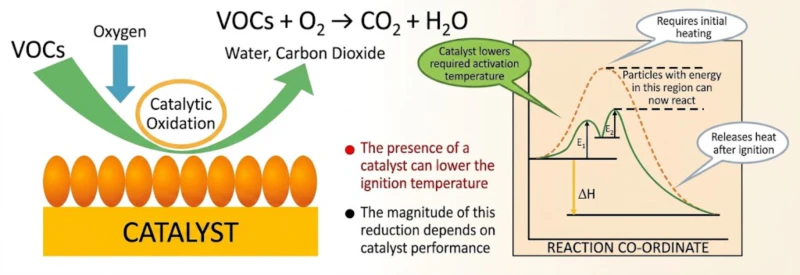
Fig 1: Molecular Activation and Exothermic Energy Feedback Loops within a Catalytic Chamber
1. High Activity: The Catalyst as a Low-Temperature Engine
Activity is the primary metric that defines a catalyst’s industrial efficiency. In the realm of VOC treatment, activity refers to the material’s ability to facilitate a chemical reaction at the lowest possible energy threshold. The technical benchmark for this is the Light-off Temperature—the point at which the catalyst initiates the destruction of organic molecules. A high-activity catalyst achieves a light-off temperature between 250 degrees Celsius and 300 degrees Celsius, which is nearly 500 degrees lower than traditional direct thermal incineration.
Kinetic Acceleration and Molecular Collision
High activity is not accidental; it is the result of precision surface engineering. By dispersing noble metals such as Platinum and Palladium across a highly porous ceramic carrier, we create an environment with a massive density of active sites. Because the carrier is engineered with high porosity, the oxygen and organic gas molecules are intimately adsorbed onto these precious metal sites. This proximity increases the statistical frequency of molecular collisions and contact time, accelerating the oxidation reaction by several orders of magnitude. For the industrial operator, this translates into a short cold-start time of only 20 to 30 minutes and the ability to maintain self-sustaining combustion using only the energy found within the waste gas itself.
In a synergistic zeolite system, high catalyst activity ensures that the concentrated stream generated during the desorption cycle is neutralized instantly. This prevents the accumulation of unreacted hazardous air pollutants and allows the overall facility to achieve a consistent removal efficiency of 95 percent or higher while keeping secondary energy costs at a minimum.
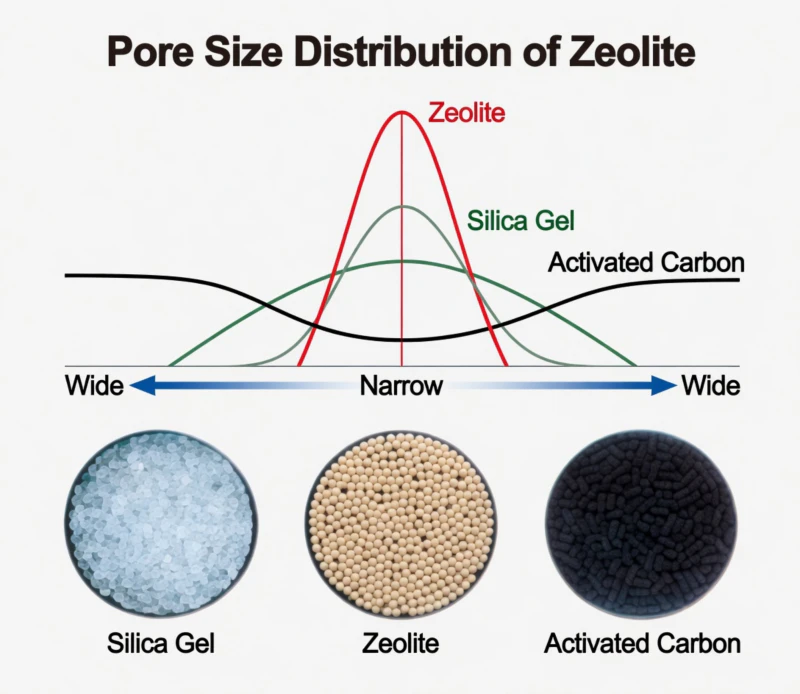
Fig 2: SEM Visualization of Porous Substrates Enabling High Active-Site Density
2. Thermal Stability: Preventing the Sintering of Active Sites
Industrial oxidation is an inherently exothermic process—it generates heat as it destroys pollution. In concentrated zeolite systems, the VOC levels in the desorption stream can reach thousands of milligrams per cubic meter, resulting in intense temperatures within the catalyst bed.
Hydrothermal Integrity and Rare-Earth Stabilization
When operating temperatures exceed 500 degrees Celsius, standard catalysts are prone to a phenomenon known as Sintering. This is a form of physical degradation where the microscopic precious metal particles begin to migrate across the surface of the carrier and aggregate into larger clusters. This reduces the effective surface area of the metal and kills the catalyst’s activity. To prevent this, industrial-grade catalysts must be engineered with exceptional thermal and hydrothermal stability.
Our premium catalyst materials incorporate rare-earth stabilizing agents that serve as molecular “anchors” for the active precious metal atoms. These anchors prevent the particles from migrating even during high-concentration exothermic surges. This ensures the catalyst maintains its ignition profile over a lifespan of several years, preventing the system from needing extra natural gas to compensate for activity loss. Proper thermal stability is the definitive factor that separates a low-cost, short-lived catalyst from a professional engineering solution that provides consistent 24/7 compliance.

Fig 3: Lattice Consistency vs. Chaotic Carbon Frameworks under Thermal Load
3. Mechanical Strength: Resilience Under High-Velocity Loads
Resisting Vibration and Thermal Shock
In large-scale industrial purification units designed to treat 200,000 cubic meters of air per hour, the physical stress on the catalyst bed is immense. High-velocity gas streams create constant aerodynamic vibration and physical abrasion against the honeycomb walls. If a catalyst carrier is fragile, it will crumble or “pulverize” over time, leading to the formation of catalyst dust. This dust not only restricts airflow—increasing the electrical load on the fan—but can also migrate downstream and contaminate other equipment.
Professional-grade industrial catalysts are manufactured with high mechanical density and resistance to thermal shock. When the system shifts from a cold standby status to its 300 degree Celsius operating state, the material expands rapidly. Only carriers with a low coefficient of thermal expansion and high structural integrity can withstand thousands of these cycles without cracking. High mechanical strength ensures that the catalyst bed remains intact, preserving the pressure drop and maximizing the energy efficiency of the facility’s ventilation network.
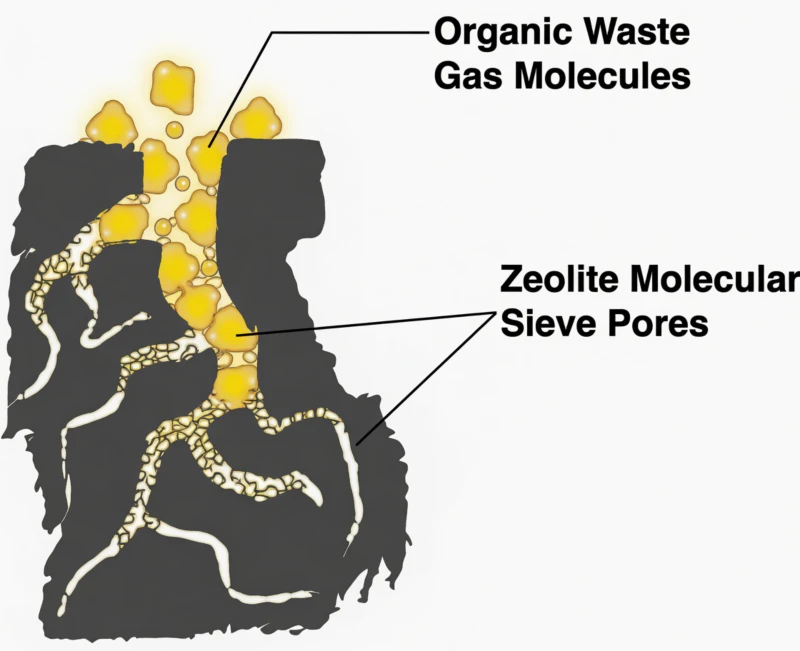
Fig 4: Catalytic Oxidizer Integration within a Cyclic Adsorption Loop
4. Service Life: The Economics of Chemical Resilience
Defending Against Catalyst Poisons
Industrial waste gases are often contaminated with “poisonous” elements such as silicon, sulfur, phosphorus, and halogens. These substances can chemically bond to the precious metal active sites, permanently masking them and effectively ending the catalyst’s useful life. High-performance catalysts are engineered with specialized surface coatings and specific co-catalysts that offer high resistance to chemical poisoning. This ensures that the system maintains its purification efficiency for several years, typically spanning between 8,000 and 12,000 hours of active operation.
Sustainability and Return on Investment
Long service life is the ultimate guarantor of your facility’s Return on Investment (ROI). Replacing a large industrial catalyst bed is a significant capital expenditure. By selecting a catalyst that combines superior chemical resilience with the thermal and mechanical durability mentioned above, facility owners minimize replacement frequency and ensure that their purification system provides continuous, high-efficiency protection for the facility’s production lines. This long-term stability is what allows enterprises to meet and exceed modern environmental sustainability targets.
Conclusion: Driving the Energy Self-Sustaining Loop
When a catalyst truly exhibits high activity, thermal stability, mechanical strength, and chemical longevity, it enables the most efficient waste gas treatment technology in existence: the Energy Self-Sustaining Loop. In this cycle, the catalyst decomposes organic pollutants and releases heat. This heat is captured by a high-efficiency heat exchanger and redirected to desorb the zeolite bed. Because the catalyst operates at such low ignition temperatures with such high efficiency, the system requires practically no external fuel after reaching its steady operating state. This creates a net-zero energy purification solution that is as safe as it is economically profitable.
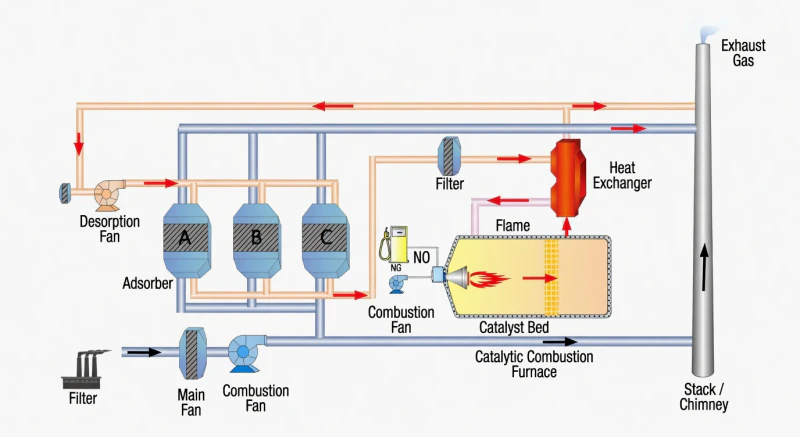
Fig 5: The Full Cyclic Synergy between Zeolite Adsorption and Catalytic Oxidation
Architect Your Compliant and Profitable Future Today
Selecting the right Heart for your catalytic combustion system is a decision that impacts environmental safety and long-term operating costs. At BAOLAN, we engineer our catalysts to survive the most rigorous industrial conditions while maintaining peak activity and thermal resilience. Contact our specialized engineering team today to architect an energy-self-sustaining VOC purification system custom-tailored to your facility’s exact solvent profile and regulatory requirements.
