In the specialized field of flue gas desulfurization (FGD), the engineering footprint of a facility is a direct physical manifestation of its internal chemical kinetics. While both the Single Alkali and Limestone-Gypsum methods aim for the total capture of Sulfur Dioxide (SO₂), they achieve this through fundamentally different molecular architectures. One relies on the lightning-fast ionic dissociation of Sodium Hydroxide (NaOH) in a purely liquid phase, while the other manages the slow, multi-stage dissolution of solid Calcium Carbonate (CaCO₃). This technical blog deconstructs these two reaction paths, exploring how molecular solubility, ionic strength, and byproduct evolution dictate the mechanical design and operational efficiency of modern industrial scrubbers.

Fig 1: Integrated Desulfurization Facility representing the Industrial Application of Liquid-Phase Kinetics
1. The Sodium Engine: Velocity through Total Solubility
The Single Alkali method utilizes Sodium Hydroxide (NaOH) or Sodium Carbonate (Na₂CO₃), reagents defined by their extreme solubility in water. From a molecular perspective, NaOH dissociates completely into Na+ and OH- ions the moment it enters the slurry. This creates a high-concentration ionic “trap” within the atomized spray droplets.
Kinetic Pathway Analysis
As the SO₂ gas makes contact with the droplet, it undergoes an instantaneous, liquid-liquid equivalent neutralization. The SO₂ hydrates to form sulfurous acid (H₂SO₃), which is immediately stripped of protons by the OH- ions to form Sodium Sulfite (Na₂SO₃). Because Na₂SO₃ remains entirely soluble, there is no “Physical Barrier” formed on the surface of the droplet. The reaction rate remains constant and high, allowing the tower to achieve SO₂ removal efficiencies of over 99% with a remarkably small footprint. This liquid-phase purity ensures the system is inherently scale-free, protecting nozzles and internal grids from the mechanical failure seen in calcium systems.
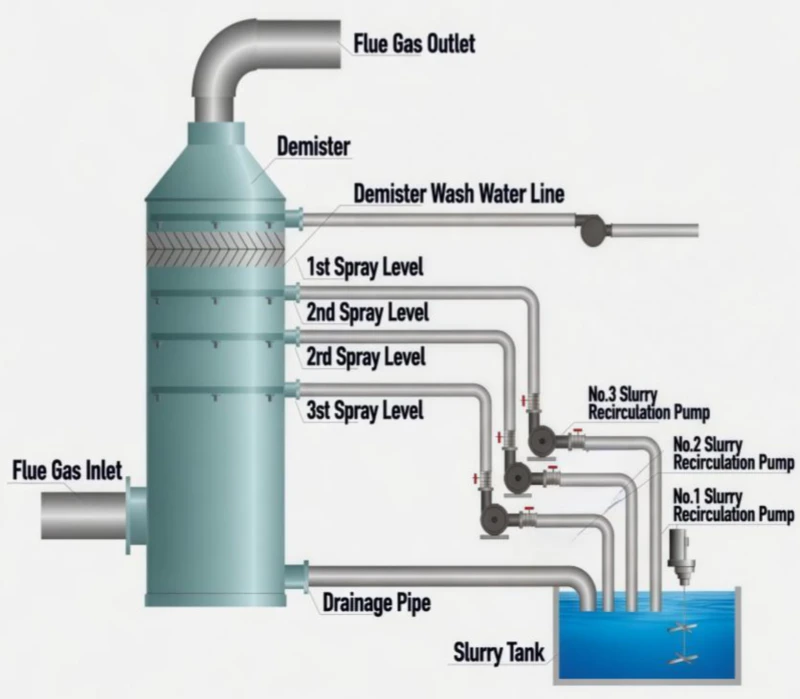
Fig 2: Liquid-Phase Process Topology for Rapid Ionic Capture
2. The Calcium Barrier: Managing Multi-Phase Dissolution
The Solid-to-Liquid Speed Limit
In contrast, the Limestone-Gypsum method utilizes Calcium Carbonate (CaCO₃), a reagent with notoriously low solubility. The reaction path is a hurdle race: gaseous SO₂ must hydrate into acid, and that acid must then attack the surface of the solid limestone particle to release Ca²⁺ ions. This dissolution is the “Rate-Limiting Step.”
Because the reaction product, Calcium Sulfite (CaSO₃), is also poorly soluble, it tends to precipitate directly onto the limestone particles, creating a “Sintering Shell” that prevents further dissolution. To overcome this molecular stubbornness, Limestone systems require massive liquid-to-gas ratios and colossal absorber towers to ensure sufficient contact time. The chemical logic here shifts from “Speed” to “Volume,” using raw material abundance to compensate for kinetic sluggishness.

Fig 3: Robust Heavy-Duty FGD Infrastructure required to manage solid-phase byproduct precipitation
3. Mechanical Adaptation: Homogenizing the Reaction Pool
Because the Limestone-Gypsum path results in heavy particulate matter, the system requires mechanical “muscle” to maintain chemical equilibrium. If the slurry pool at the base of the tower is left stagnant, the calcium sulfite will settle into a concrete-like sediment, leading to catastrophic scaling.
Hydrodynamic Turbulence for Kinetic Continuity
Side-entry agitators are mandatory for calcium systems. These units create intense internal turbulence, keeping the solid limestone and its reaction byproducts in a homogeneous suspension. This constant mechanical stirring ensures that the oxidation air (pumped by Roots blowers) can penetrate the liquid and reach the sulfite molecules, converting them into stable Gypsum ($CaSO_4 \cdot 2H_2O$). In contrast, Single Alkali (NaOH) systems require significantly less agitation power because their byproducts are inherently soluble, allowing for a far leaner mechanical profile.

Fig 4: Heavy-Duty Mechanical Agitator ensuring Solid Suspension in CaCO3 Systems
4. Protecting the Stack: Managing the Wet Exhaust Path
Both pathways result in a saturated flue gas stream laden with microscopic liquid aerosols. However, the “threat level” of these droplets differs based on the formula. In the NaOH path, the droplets contain soluble sodium salts. In the CaCO₃ path, they contain abrasive gypsum and limestone particulates.
Inertial Separation Dynamics
High-efficiency demisters at the tower outlet utilize corrugated blade geometry to force the gas stream through rapid, repetitive direction changes. While the gas easily navigates these turns, the heavier liquid droplets collide with the blades due to inertia. In Limestone systems, these demisters must be equipped with powerful automated wash systems to prevent the abrasive solids from forming a hard crust on the blades, which would otherwise restrict airflow and increase operational costs.
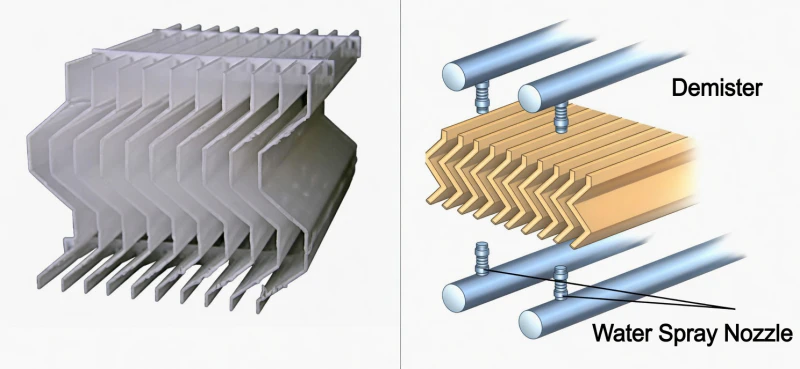
Fig 5: Corrugated Blade Demisting Module with Automated Flushing Grid
5. Selection Strategy: ROI vs. Kinetic Purity
Selecting between NaOH and CaCO₃ paths is a decision of both environmental science and financial foresight. For mega-scale facilities, the Limestone-Gypsum path remains the economic leader due to the ultra-low cost of raw limestone and the ability to monetize निर्माण-grade gypsum as a byproduct. However, this comes at the cost of high maintenance and a massive engineering footprint.
For the “Lean Industry” sector—semiconductors, high-tech manufacturing, and urban-based metallurgy—the Single Alkali (NaOH) pathway is the definitive winner. Its lightning-fast kinetics allow for absorber towers that are 40% smaller than their calcium counterparts, with 0% risk of hard-scaling. By driving outlet emissions consistently below 35 mg/Nm3 through purely ionic reactions, enterprises can achieve total regulatory peace of mind without the operational headache of managing solid-phase residue.
Architect Your Compliant Future Today
Don’t let complex flue gas profiles or hard-scaling risks compromise your facility’s environmental roadmap. Implement the power of molecular-grade desulfurization to ensure safe, stable, and economically superior flue gas purification. Contact BAOLAN EP INC. today to design a specialized desulfurization loop custom-tailored to your facility’s exact volumetric and sulfur concentration targets.
