In the modern landscape of high-precision manufacturing, environmental compliance is often viewed as a “cost center.” Traditional Volatile Organic Compound (VOC) treatment methods, specifically direct thermal combustion, are notorious for their voracious appetite for natural gas. When faced with large airflows and low pollutant concentrations—typical of electronics, printing, and automotive coating lines—direct combustion becomes a financial burden that can erode a facility’s entire profit margin. However, the emergence of Zeolite Adsorption-Concentration + Catalytic Combustion has turned this paradigm on its head. With operational costs reaching as low as 5 percent of traditional methods, the “Energy-Saving Secret” lies in a sophisticated combination of molecular physics, thermodynamic feedback, and crystalline geometry.

Fig 1: Large-Scale Industrial Integration: High Purification Rates at a Fraction of the Cost
1. The Dilution Trap: Why Direct Combustion Fails
The primary challenge in industrial VOC abatement is not the toxicity of the solvents, but their dispersion. Direct thermal combustion requires heating the entire exhaust air stream—often reaching 100,000 $m^3/h$ or more—to a temperature of at least 800 degrees Celsius to achieve oxidative destruction. When the VOC concentration is low (e.g., less than 500 $mg/m^3$), the pollutants themselves do not provide enough “fuel” to maintain these temperatures.
The Auxiliary Fuel Nightmare
In this scenario, a direct combustion system acts as a giant radiator for the atmosphere, burning massive amounts of expensive natural gas simply to heat clean air. This results in “Negative Energy Returns,” where the energy cost of purification exceeds the value of the manufacturing process itself. Furthermore, high-temperature combustion inevitably produces Nitrogen Oxides (NOx), requiring additional treatment and creating a cycle of secondary pollution and secondary costs.
Zeolite systems solve this by refusing to play the game of “heating the air.” Instead, they treat the exhaust air as a temporary carrier, using molecular sieves to harvest only the VOC molecules and letting the clean, cold air return to the atmosphere immediately.
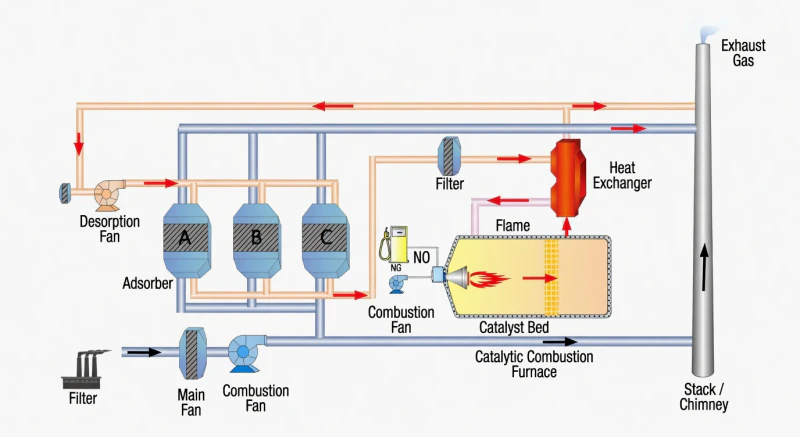
Fig 2: The Synergistic Loop: Isolating Pollutants from the Thermal Load
2. Concentration Spiking: Turning Liability into Fuel
The 20:1 Enrichment Ratio
The “Secret” begins with the Desorption Phase. As the zeolite molecular sieve captures VOCs, it becomes saturated. The system then switches to a regeneration mode using a high-temperature hot air stream. However, this desorption airflow is typically only 1/10th to 1/20th the volume of the original raw exhaust.
By detaching the VOCs into a much smaller volume of air, the concentration of organic pollutants spikes by 10 to 20 times. For example, a dilute stream of 200 $mg/m^3$ is concentrated into a dense stream of 4,000 $mg/m^3$. At this density, the VOCs transition from a toxic waste into a high-energy fuel. When this concentrated gas enters the catalytic oxidizer, the energy released by its destruction is so intense that the system becomes thermally self-sufficient.
Operational Savings: Because the combustion energy is provided by the pollutants themselves, the need for external natural gas is eliminated during steady-state operation, reducing the fuel cost to zero.
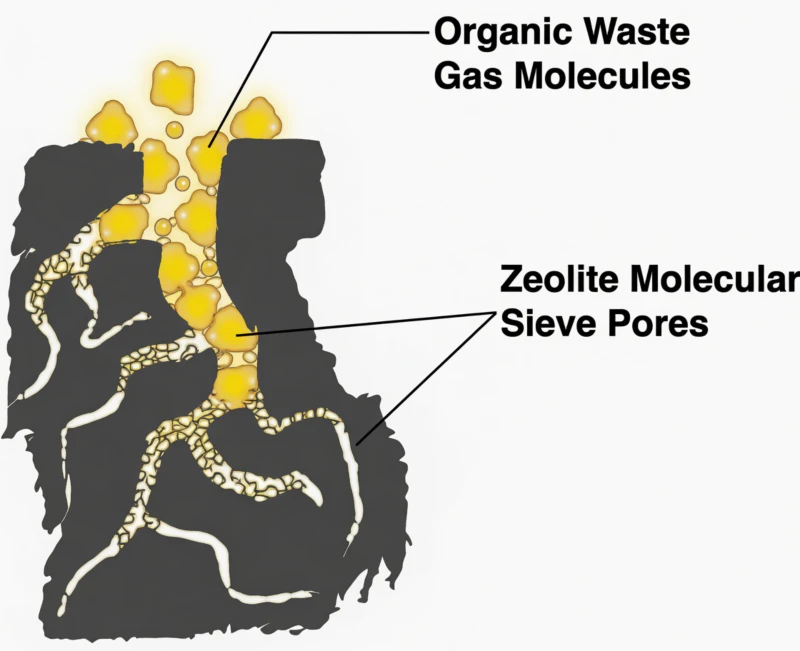
Fig 3: Molecular Enrichment: Spiking VOC density to enable self-sustaining oxidation
3. Catalytic Combustion vs. Thermal Incineration
Bridging the 500-Degree Temperature Gap
The second major energy-saving secret lies in the ignition temperature. Direct thermal combustion is a “brute force” process, requiring 800 degrees Celsius to crack organic bonds. Catalytic combustion, utilizing high-activity precious metal beds, lowers the activation energy of the reaction. This allows VOCs to be completely oxidized at a mere 250 to 300 degrees Celsius.
Maintaining 300 degrees Celsius requires exponentially less energy than maintaining 800 degrees Celsius. In a Zeolite system, this “Low-Temperature Oxidation” is coupled with an internal high-efficiency heat exchanger. The exothermic heat generated by the flameless combustion of concentrated VOCs is harvested and fed back to pre-heat the incoming gas. This thermal feedback loop creates a self-sustaining cycle where the destruction of pollutants provides the energy to destroy more pollutants. The resulting products—harmless carbon dioxide and water vapor—exit the system at a low enough temperature that heat loss is minimized, maximizing total system efficiency.
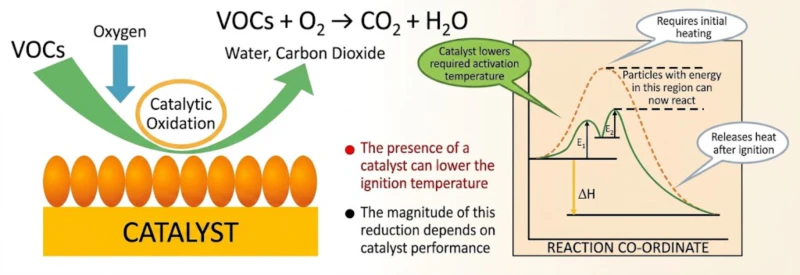
Fig 4: Low-Temperature Oxidation Mechanism and Exothermic Feedback
4. Micro-Engineering: Reducing Electrical Resistance
Energy saving is not only about gas consumption; it is also about electricity. In large-scale industrial ventilation, the “Wind Resistance” or pressure drop across a filtration system determines the power requirement of the induction fans. Standard media like disordered activated carbon creates a chaotic and high-resistance pathway for air, forcing fans to work harder and consume more power.
The BAOLAN Zeolite system utilizes a **Honeycomb Crystalline Framework**. Through Scanning Electron Microscopy, it is evident that zeolite channels are perfectly straight and uniformly aligned. This ordered geometry allows massive airflows—up to 200,000 $m^3/h$—to pass through the bed with a resistance of only 300 Pa. By maintaining an empty tower wind speed of 0.8 to 1.5 m/s, the system minimizes aerodynamic turbulence.
Fluid Dynamic Advantage: Lower wind resistance translates directly into lower fan amperage, reducing the monthly electrical bill for exhaust management by 30 to 50 percent compared to traditional particulate-heavy or dense-bed systems.

Fig 5: Geometric Efficiency: Regular Honeycomb Channels Minimizing System Pressure Drop
The 1/20th Verdict: A Sustainable ROI
The cumulative result of these engineering secrets is a profound shift in operational expenditure. When we compare a standard 50,000 $m^3/h$ exhaust stream with a low VOC concentration, the direct thermal combustion method would cost tens of thousands of dollars per month in natural gas. The Zeolite Adsorption-Concentration + Catalytic Combustion system reduces this to a mere fraction. By concentrating the gas, lowering the ignition temperature, and harvesting the reaction heat, the energy cost is effectively reduced to 1/20th of the thermal alternative.
Safety and Stability
Beyond energy, the inorganic zeolite matrix is non-flammable and thermally stable. It eliminates the catastrophic fire risks associated with activated carbon beds handling ketones or alcohols.
Long Service Life
Premium catalysts with high activity and anti-poisoning properties ensure the system provides 8,000 to 12,000 hours of peak performance before media maintenance is required.
Self-Sustaining Loop
Once the catalyst reaches ignition temperature, the oxidation of concentrated VOCs produces enough heat to maintain the desorption process without external fuel.
By adhering to ISO9001 quality management and leveraging state-of-the-art material science, industrial facilities can now achieve environmental excellence without compromising their financial health. The secret to the 1/20th operational cost is not just one component—it is the holistic synergy of concentration, low-temperature catalysis, and aerodynamic micro-engineering.
Unleash the Power of Energy-Neutral Purification
Don’t let high energy costs and safety risks compromise your facility’s environmental roadmap. Implement the power of cyclic zeolite technology to ensure safe, stable, and economically superior VOC purification. Whether you are managing the delicate solvents of a semiconductor plant or the massive air volumes of a commercial printing line, our custom-engineered adsorption-combustion loops provide the definitive answer. Contact our expert engineering team today to architect a system custom-tailored to your exact solvent profile and sustainability goals.
