In industrial emission control, Sulfur Dioxide (SO₂) receives the majority of regulatory attention. However, for facility managers and maintenance engineers, the true threat lies in its highly corrosive derivative: Sulfur Trioxide (SO₃). When flue gas cools, SO₃ reacts with moisture to form a deadly Sulfuric Acid Mist—a silent assassin that aggressively attacks baghouse filters, induced draft fans, and stack infrastructure, leading to catastrophic equipment failure and the infamous “blue plume” emissions. Traditional wet scrubbers often fail to capture these sub-micron acid aerosols effectively. Enter the Sodium Bicarbonate Dry Desulfurization (SDS) system. By leveraging the hyper-reactivity of thermally activated sodium carbonate, the SDS process provides unparalleled synergistic control, neutralizing SO₃ in the dry gas phase before it can ever condense. This technical breakdown explores how sodium-based dry kinetics transform a severe corrosion liability into a stable, harmless powder.

Fig 1: Industrial Deployment of the BLSDS Series Dry Desulfurization Architecture
1. The Acid Dew Point: Anatomy of a Corrosion Crisis
To understand the protective value of the SDS system, one must first analyze the thermodynamics of Sulfur Trioxide (SO₃). In high-temperature industrial kilns, incinerators, and boilers, approximately 1% to 5% of the total SO₂ generated is naturally oxidized into SO₃. While it constitutes a minor percentage of the total volume, its physical behavior in the exhaust duct makes it disproportionately destructive.
The Condensation Trap
SO₃ possesses a notoriously high “Acid Dew Point”—typically ranging between 120°C and 150°C depending on moisture content. As the hot flue gas travels through downstream ductwork and approaches the baghouse filter, it inevitably loses thermal energy. The moment the temperature drops below this critical dew point, gaseous SO₃ reacts with water vapor to condense into highly concentrated droplets of liquid sulfuric acid (H₂SO₄). This sticky, highly corrosive mist immediately coats the internal surfaces of all downstream equipment.
Traditional wet limestone scrubbers are often positioned downstream of the baghouse and operate at low temperatures, doing nothing to protect the filter bags from this upstream condensation. Furthermore, wet scrubbers struggle to capture these sub-micron acid aerosols, allowing them to pass through the stack and form a highly visible, highly regulated “blue plume” in the atmosphere.
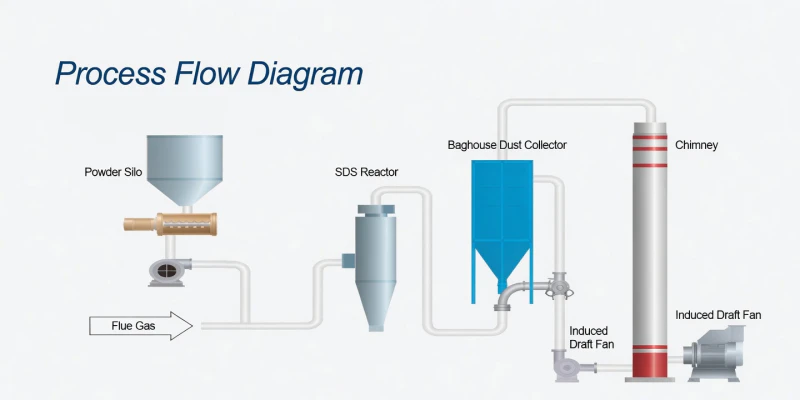
Fig 2: Strategic Injection: Neutralizing Acid Gases Upstream of Sensitive Filtration Assets
2. The Sodium Solution: Thermal Activation Kinetics
The “Popcorn Effect” and Molecular Reactivity
The SDS system solves the SO₃ crisis by eradicating the acid in its gaseous phase, well before it reaches the dew point. The process relies on pneumatically injecting ultra-fine Sodium Bicarbonate (NaHCO₃) powder directly into the high-temperature flue gas duct (typically operating between 140°C and 260°C).
When exposed to this intense thermal energy, the sodium bicarbonate undergoes an instant endothermic decomposition, transforming into Sodium Carbonate (Na₂CO₃), Carbon Dioxide, and water vapor. As the CO₂ escapes from within the solid particle, it shatters the crystalline structure, creating a vast network of microscopic pores. This “popcorn effect” results in a highly activated, highly porous sodium carbonate molecule with an immense specific surface area.
Because sodium is significantly more reactive than calcium-based absorbents, this highly porous Na₂CO₃ hunts down and neutralizes not only SO₂, but aggressively bonds with trace amounts of SO₃ to form stable, solid Sodium Sulfate (Na₂SO₄) and Carbon Dioxide.
Synergistic Reaction Pathways
Phase 1: Thermal Decomposition
2NaHCO₃ + Heat → Na₂CO₃ + CO₂↑ + H₂O
Phase 2: Acid Mist (SO₃) Eradication
Na₂CO₃ + SO₃ → Na₂SO₄ + CO₂↑
Phase 3: Primary Desulfurization
Na₂CO₃ + SO₂ → Na₂SO₃ + CO₂↑
3. The Filter Cake: The Ultimate Baghouse Shield
Baghouse filters are notoriously vulnerable to sulfuric acid mist. When acid condenses on the filter bags, it causes rapid chemical hydrolysis of the fabric (especially PPS and PTFE materials) and creates a wet, sticky mud with the fly ash. This phenomenon, known as “bag blinding,” results in unmanageable pressure drops and catastrophic filter failure.
Alkaline Crust Formation
The SDS system completely reverses this vulnerability. As the gas stream flows from the duct into the baghouse, it carries a significant amount of highly reactive, unreacted Sodium Carbonate powder. This alkaline powder is continuously deposited onto the surface of the filter bags, forming a porous, highly basic “Filter Cake.”
As the flue gas is forced to pass through this basic crust, any residual SO₃ molecules that escaped the pipeline reaction are forced into intimate contact with the sodium carbonate. The acid is instantly neutralized directly on the surface of the bag. Instead of forming a sticky, destructive acid mud, the byproduct is dry, powdery Sodium Sulfate, which is easily dislodged during the automated pulse-jet cleaning cycle. This synergistic mechanism actively shields the fragile fabric fibers from acid hydrolysis, preserving the integrity of the filtration system.
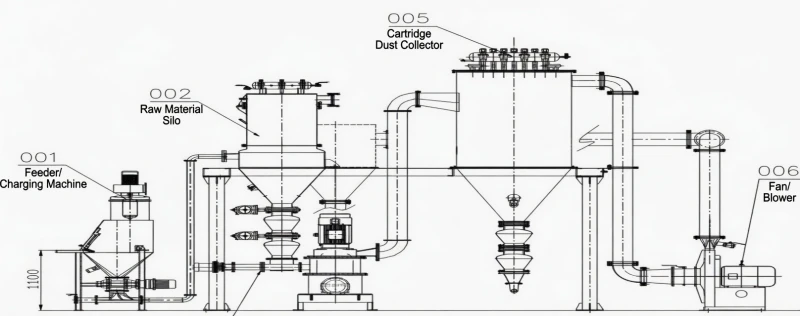
Fig 3: Sub-Micron Pulverization ensuring an even, highly porous alkaline filter cake
4. Asset Protection: Securing the Downstream Flow
The protective umbrella of the SDS system extends far beyond the baghouse. By entirely eliminating sulfuric acid mist from the exhaust profile, facility managers secure the structural integrity of the most expensive aerodynamic assets in the plant.
Induced Draft (ID) Fan Longevity
The ID fan operates under immense mechanical stress. When acid mist passes through the fan, it condenses on the high-speed impeller blades, causing aggressive pitting, severe corrosion, and eventually, catastrophic rotor imbalance. Because the SDS process captures all SO₃ before the baghouse, the gas passing through the ID fan is entirely dry and free of acidic aerosols. This allows the use of standard carbon steel impellers, completely bypassing the need for ultra-expensive, corrosion-resistant alloy materials or frequent impeller replacements.
Eliminating the “Blue Plume”
Sub-micron sulfuric acid aerosols are highly effective at scattering sunlight, creating a highly visible, highly regulated “blue plume” at the exit of the stack—even if standard SO₂ monitors read zero. Furthermore, acid condensation inside the stack structure causes structural degradation over time. The synergistic SO₃ elimination of the SDS system ensures that the final discharge is an invisible, dry, and entirely benign exhaust, guaranteeing both structural safety and perfect visual compliance.
Maximize Your Equipment’s Lifecycle Today
Don’t let invisible sulfuric acid mist compromise your filtration systems, destroy your aerodynamic infrastructure, or trigger regulatory scrutiny. Implementing the BAOLAN SDS Dry Desulfurization system is an investment in absolute asset protection. Turn your pipeline into a high-speed chemical reactor and secure your operational continuity. Contact our specialized engineering team today to design a completely dry, zero-corrosion emission control architecture for your facility.
